
Furthermore, high-energy physics research can create an even more energetic state of matter, the so-called quark-gluon-plasma. In particular, several particle detectors rely on phase transitions. When the free energy exhibits continuous first derivatives but discontinuous second derivatives, the phase transition is called second order. Phase transitions often occur in nature, but they are also used in many technologies. The solid-liquid-gas phase transition of most substances is first order. When the first derivative of the free energy with respect to one of its dependent thermodynamic variables is discontinuous across a phase transition, this is an example of what is called a first order phase transition. First, although the free energy is continuous across the phase transition, its first derivative, \(\partial G/\partial T\) is not: The slope of \(G(T)\) in the solid region is different from the slope in the liquid region. \(T\) between the solid and liquid phases of benzene: We immediately notice several things. Figure 13.4: Some of the ice polymorphs.Ĭonsider, first, the behavior of \(G\) vs. We review the effects of excited-state quantum phase transitions (ESQPTs) in interacting many-body systems with finite numbers of collective degrees of freedom. 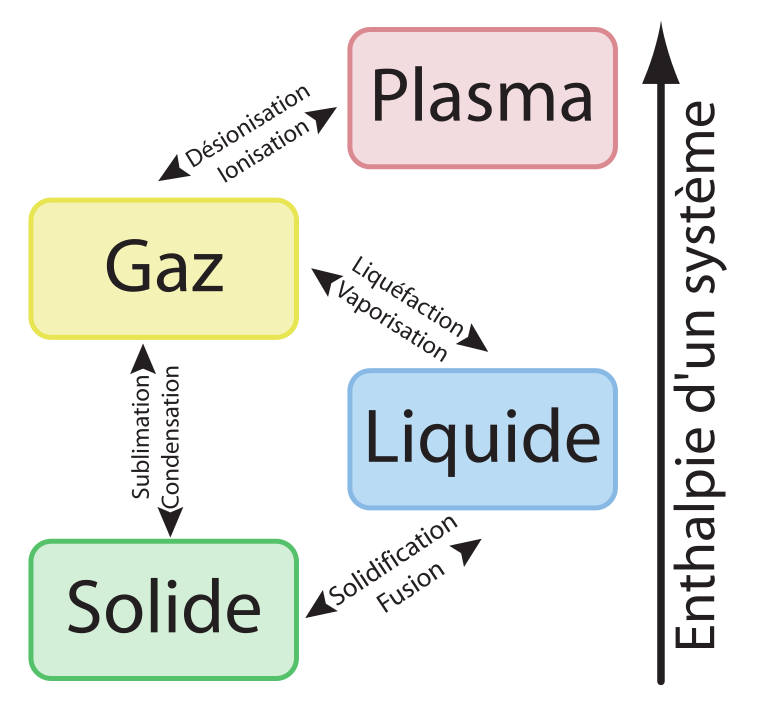
The behavior of \(G(N, P,T)\), particularly as a function of \(P\) and \(T\), can signify a phase transition and can tell us some of the thermodynamic properties of different phases. The Gibbs free energy is a particularly important function in the study of phases and phase transitions. The figure below shows just a few of the known ice polymorphs (more were discussed in class): Figure 13.3: Phase diagram of water. Again, however, if the pressure axis is expanded, these show up. In article number 2300623, Junhua Hu and co-workers, report that a core shell structure of (Co x Fe 1x) 3 N with high dispersibility is induced by an intermediate phase transition process. As with benzene, the solid region of the phase diagram of water also exhibits a rich variety of different structures, the ice polymorphs, not shown in the phase diagram above. The synergistic effects of phase transition and electron-spin regulation boosts oxygen electrocatalysis. The water phase diagram is shown in the figure below: Here we see the slight slope to the left of the solid-liquid coexistence line. C\), its freezing point at standard pressure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
